The Challenge
The adeno-associated viral (AAV) vector appears to be the safest and most effective vehicle, allowing for long-term gene and protein expression after a single injection. There are 12 naturally occurring serotypes (AAV-1 to AAV-12) and over 100 variations of AAV, each having slightly different amino acid sequences, particularly within the hypervariable portions of the capsid proteins, and hence slightly different gene delivery capabilities.
Challenges faced-
- Identification of various players working for the treatment of rare disease Hemophilia A using AAV-based gene therapy.
- Key Serotypes/Promoters/Regulatory elements that form part of the genetic construct for AAV-based therapy
- Identification of various therapeutic candidates in clinical and pre-clinical stages
- Identification of potential partners for AAV-based gene therapy in Hemophilia A.
Our Solution
Our team formulated a comprehensive strategy to conduct the study. Our scope covered clinical and pre-clinical research areas and in-depth analysis to generate actionable insights for our clients. We identified emerging trends for AAV-based Gene Therapy, including regulatory elements explored in constructing an expression cassette for a practical therapeutic approach.
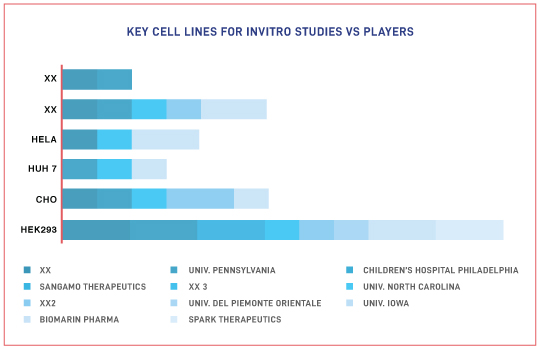
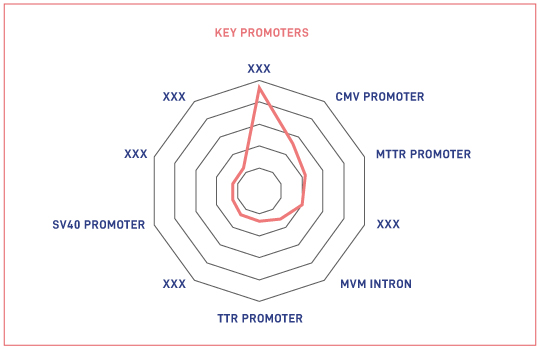
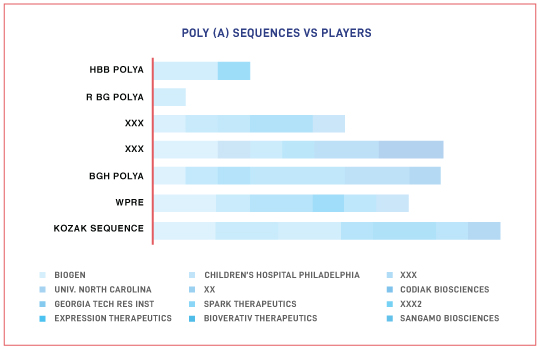
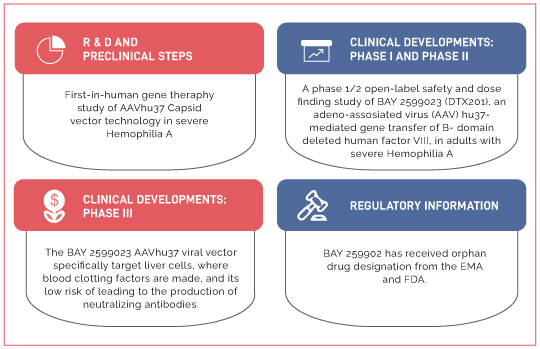
The Impact
Our in-depth analysis of therapeutic candidates under various stages of development and correlating with their pre-clinical/ R&D focus of other players helped our client understand of key genetic constructs and their impact on gene therapy and performance on animal models. Furthermore, the Identification of emerging trends and comparative analysis of the R&D focus of key players in the domain proved to be valuable in making key decisions pertaining to their therapeutic portfolio and in improvising their research focus.

